Signals from nociceptors
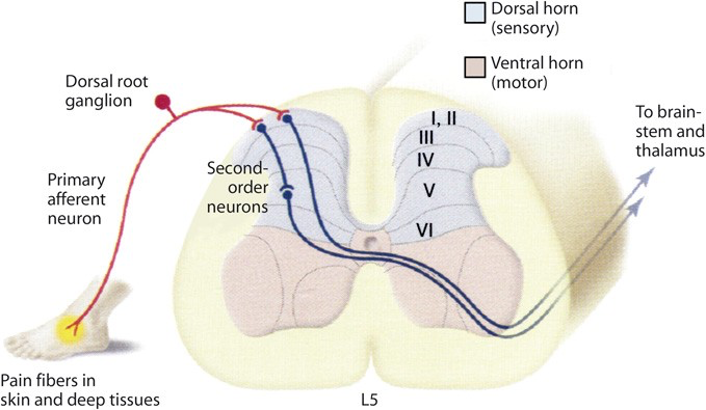
Signals from nociceptors are conveyed to neurons in the dorsal horn of the spinal cord. The perception of noxious stimuli arises from signals in the peripheral axonal branches of nociceptive sensory neurons whose cell bodies are located in dorsal root ganglia or the trigeminal ganglia. The central branches of these neurons terminate in the spinal cord in a highly orderly manner. Most terminate in the dorsal horn. Primary afferent neurons that convey distinct sensory modalities terminate in different laminae.
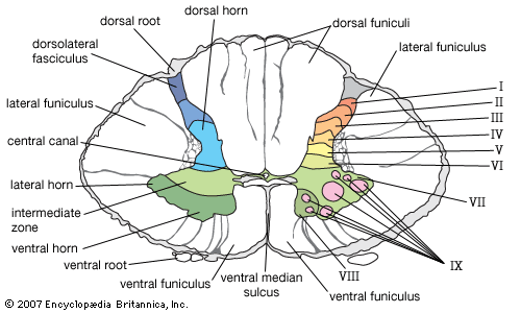
Thus, there is a tight link between the anatomical organization of dorsal horn neurons, their receptive properties, and their function in sensory processing. Many neurons in the most superficial lamina of the dorsal horn, termed lamina I or the marginal layer, respond to noxious stimuli conveyed by Adelta and C fibers. Because they respond selectively to noxious stimulation they have been called nociception-specific neurons. This set of neurons projects to higher brain centers, notably the thalamus. A second class of lamina I neurons receives input from C fibers that are activated selectively by intense cold. Other classes of lamina I neurons respond in a graded fashion to both innocuous and noxious mechanical stimulation and thus are termed wide-dynamic-range neurons.
Lamina II, the substantia gelatinosa, is a densely packed layer that contains many different classes of local interneurons, some excitatory and others inhibitory. Some of these interneurons respond selectively to nociceptive inputs, whereas others also respond to innocuous stimuli. Laminae III and IV contain a mixture of local interneurons and supraspinal projection neurons. Many of these neurons receive input from Abeta afferent fibers that respond to innocuous cutaneous stimuli, such as the deflection of hairs and light pressure. Lamina V contains neurons that respond to a wide variety of noxious stimuli and project to the brain stem and thalamus. These neurons receive direct inputs from Abeta and Adelta fibers and, because their dendrites extend into lamina II, are also innervated by C fibers nociceptors.
Neurons in lamina V also receive input from nociceptors in visceral tissues. The convergence of somatic and visceral nociceptive inputs onto individual lamina V neurons provides one explanation for a phenomenon called “referred pain,” a condition in which pain from injury to a visceral tissue is perceived as originating from a region of the body surface. Patients with myocardial infarction, for example, frequently report pain from the left arm as well as the chest.
This phenomenon occurs because a single lamina V neuron receives sensory input from both regions, and thus a signal from this neuron does not inform higher brain centers about the source of the input. As a consequence, the brain often incorrectly attributes the pain to the skin, possibly because cutaneous inputs predominate. Another anatomical explanation for instances of referred pain is that the axons of nociceptive sensory neurons branch in the periphery, innervating both skin and visceral targets. Neurons in lamina VI receive inputs from large diameter fibers that innervate muscles and joints. These neurons are activated by innocuous joint movement and do not contribute to the transmission of nociceptive information. In contrast, many neurons located in laminae VII and VIII, the intermediate and ventral regions of the spinal cord, do respond to noxious stimuli.
These neurons typically have complex response properties because the inputs from nociceptors to these neurons are conveyed through many intervening synapses. Neurons in lamina VII often respond to stimulation of either side of the body, whereas most dorsal horn neurons receive unilateral input. The activation of lamina VII neurons is therefore thought to contribute to the diffuse quality of many pain conditions.
Nociceptive sensory neurons that activate neurons in the dorsal horn of the spinal cord release two major classes of neurotransmitters. Glutamate is the primary neurotransmitter of all primary sensory neurons, regardless of sensory modality. Neuropeptides are released as cotransmitters by many nociceptors with unmyelinated axons. These peptides include substance P, calcitonin gene–related peptide (CGRP), somatostatin, and galanin. Glutamate is stored in small, electron-lucent vesicles, whereas peptides are sequestered in large, dense-core vesicles at the central terminals of nociceptive sensory neurons. Separate storage sites permit these two classes of neurotransmitters to be released under different physiological conditions. Of the neuropeptides expressed by nociceptive sensory neurons, the actions of substance P, a member of the neurokinin peptide family, have been studied in most detail. Substance P is released from the central terminals of nociceptive afferents in response to tissue injury or after intense stimulation of peripheral nerves.
Its interaction with neurokinin receptors on dorsal horn neurons elicits slow excitatory postsynaptic potentials that prolong the depolarization elicited by glutamate. Although the physiological actions of glutamate and neuropeptides on dorsal horn neurons are different, these transmitters act coordinately to regulate the firing properties of dorsal horn neurons.
There is no efficient means for peptide reuptake into nerve terminals, so neuropeptides released from sensory terminals diffuse over a greater distance than glutamate and thus have excitatory influences on many dorsal horn neurons in the immediate vicinity of the release site. This diffuse signaling system may contribute to the poorly localized character of many pain conditions. In addition, the levels of neuropeptide expression in primary nociceptive neurons are elevated in some pathological conditions. Such reactive changes in peptide expression may contribute to the enhanced excitability of dorsal horn neurons that accompanies some chronic pain states.
Details of the interaction of neuropeptides with their receptors on dorsal horn neurons have suggested strategies for pain regulation. Infusion of substance P coupled to a neurotoxin into the dorsal horn of experimental animals results in selective destruction of neurons that express neurokinin receptors. Animals treated in this way fail to develop the central sensitization that is normally associated with peripheral injury.